Introduction
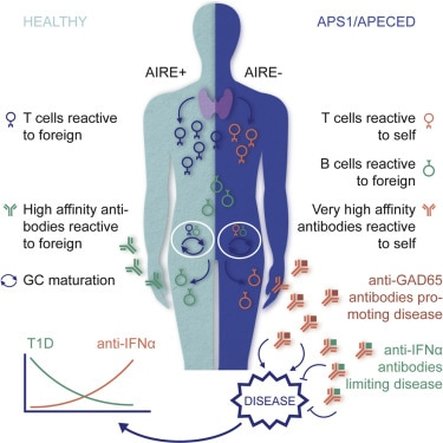 The above image shows the loss of self-tolerance in APECED patients. Without AIRE, self-reactive T cells are able to enter the body and cause autoimmune diseases.
The above image shows the loss of self-tolerance in APECED patients. Without AIRE, self-reactive T cells are able to enter the body and cause autoimmune diseases.
APECED is a rare autoimmune disorder caused by a general loss of self-tolerance, meaning the immune cells attack the patient’s body [1]. Normally, the body randomly generates new immune cells, called T-cells, that each are reactive to a specific peptide. However, because some of these new T-cells are reactive against self-peptides, all new T-cells go through a selection process in the thymus, a primary lymphoid organ located in the chest. During this selection process, self-tolerance is created through the elimination of any T-cells that bind to self-peptides. This process results in set of adaptive immune cells that are ready to defend the body against foreign invaders (viruses, bacteria, etc.) but are self-tolerant, and therefore will not attack the person’s body. In APECED patients, this crucial selection process does not occur, meaning all of the newly generated T-cells, both self-reactive and self-tolerant, are released into the body which results in a number of autoimmune disorders.
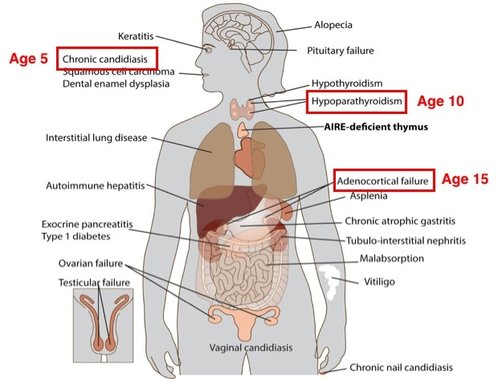
The symptoms associated with APECED can vary by patient, but in a clinical setting, APECED is diagnosed based on the presence of two out of the following three symptoms: chronic mucocutaneous candidiasis which is yeast infection of the skin and mucosa [1]; hypoparathyroidism, an endocrine disorder causing muscle cramps/spasms, fatigue and weakness, and headaches [2]; and adrenocortical insufficiency chronic fatigue, muscle weakness, loss of appetite, abdominal pain and weight loss [3].
Disease Mechanism
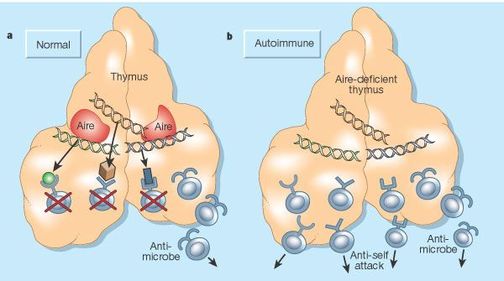
APECED is caused by a loss of function of a transcription factor called the AutoImmune REgulator (AIRE). Without AIRE, the selection process in the thymus cannot eliminate self-reactive T-cells. AIRE’s role in the selection process is to induce the transcription of self-peptides, called self-antigens, which are then presented to new T-cells in the thymus. Any T-cell that tightly binds the self-antigen receives apoptosis signals, resulting in the destruction of all self-reactive T-cells. This process creates a self-tolerant immune system, because the remaining T-cells will only attack foreign antigens.
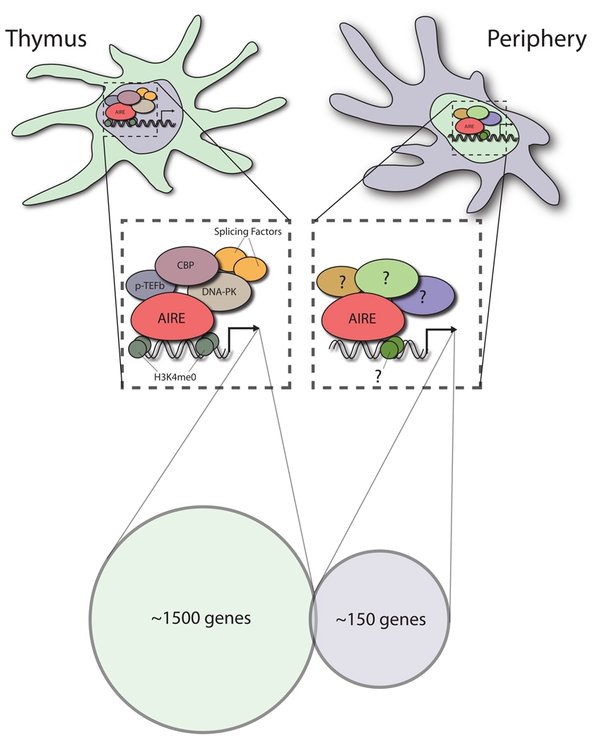
As a transcription factor, AIRE mainly functions in the nucleus where it induces the expression of these self-antigens. AIRE forms complexes with many other proteins, including histone binding proteins, transcription elongation factors, and DNA binding proteins to carry it its function as a transcriptional activator [4]. AIRE is well conserved among mammals, which have similar immune systems, but AIRE homologs in other model organisms, including the zebrafish, have a lower percent identity because their immune systems differ from mammals.
Specific Aims
Recent studies have identified extra-thymic AIRE expressing cells in secondary lymphoid organs, such as lymph nodes, that are thought to play a role in maintaining peripheral self-tolerance. In eTACs, similarly to in the thymus, AIRE promotes the expression of self-antigens. However, the self-antigens expressed in eTACs differ from those in the thymus. Therefore, the primary goal of this study is to better understand the mechanism by which AIRE promotes the expression of unique self-antigens in eTACs.
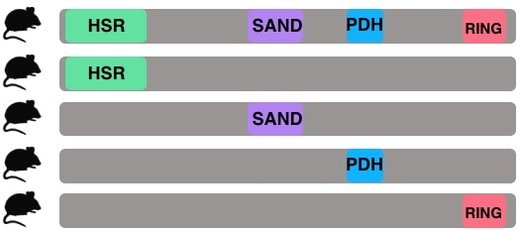
Aim 1: Which AIRE domains are sufficient for self-antigen expression in eTACs?
The purpose of aim 1 is to understand the roles of each individual AIRE domain in eTACs. Because it is already known that eTACs express different self-antigens than are in the thymus, exploring the function of individual AIRE domains in eTACs could shed light on why the self-antigens in eTACs differ. This can be studied by comparing eTACs from mice with single domain AIRE constructs to wild type mice. By tagging AIRE with GFP in all mice, fluorescence activated cell sorting can be used to isolate AIRE expressing cells. Then, RNA-seq analysis will be performed to analyze the self-antigen expression in wild type versus the single-domain construct AIRE expressing cells. My hypothesis is that the wild type eTACs will have the highest expression of self-antigens because they have the full AIRE protein. Additionally, I expect that the PDH domain will have higher self-antigen expression than the other domains because PDH has been shown to be an activation domain [5].
The purpose of aim 1 is to understand the roles of each individual AIRE domain in eTACs. Because it is already known that eTACs express different self-antigens than are in the thymus, exploring the function of individual AIRE domains in eTACs could shed light on why the self-antigens in eTACs differ. This can be studied by comparing eTACs from mice with single domain AIRE constructs to wild type mice. By tagging AIRE with GFP in all mice, fluorescence activated cell sorting can be used to isolate AIRE expressing cells. Then, RNA-seq analysis will be performed to analyze the self-antigen expression in wild type versus the single-domain construct AIRE expressing cells. My hypothesis is that the wild type eTACs will have the highest expression of self-antigens because they have the full AIRE protein. Additionally, I expect that the PDH domain will have higher self-antigen expression than the other domains because PDH has been shown to be an activation domain [5].
Aim 2: Does AIRE mediated self-antigen expression change over time in eTACs?
In APECED patients, symptoms do not begin showing up until around age 5 and continue to present throughout teenage years and even later in life. Given this, the goal of this aim is to determine whether AIRE’s role in eTACs changes over time. To test this question, the self-antigen expression in eTACs of wild type mice of three different age groups will be analyzed. The ages are: 1 month to represent teenage mice, 5 months to represent mature adults, and 10 months to represent middle aged [6]. RNA-seq will be used to analyze self-antigen expression in eTACs.
In APECED patients, symptoms do not begin showing up until around age 5 and continue to present throughout teenage years and even later in life. Given this, the goal of this aim is to determine whether AIRE’s role in eTACs changes over time. To test this question, the self-antigen expression in eTACs of wild type mice of three different age groups will be analyzed. The ages are: 1 month to represent teenage mice, 5 months to represent mature adults, and 10 months to represent middle aged [6]. RNA-seq will be used to analyze self-antigen expression in eTACs.
|
My hypothesis is that self-antigen expression in eTACs will increase with age, meaning the oldest mice will have the highest self-antigen expression. The types of self-antigens generally expressed by AIRE in eTACs include hormones, cell adhesion proteins, and musical proteins, but given that AIRE self-antigen expression is stochastic, I don’t expect the proportions of self-antigen types to change.
|
|
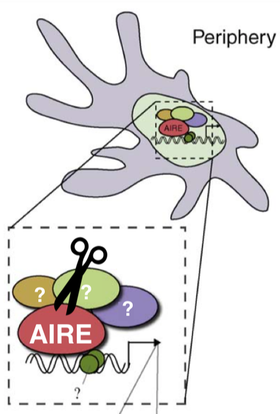
Aim 3: Which proteins interact with AIRE in eTACs?
While it is known that eTACs express self-antigens that differ from the thymus, it is not known what proteins AIRE interacts with in eTACs. Uncovering proteins that work with AIRE to promote self-antigen expression is important for understanding why self-antigen expression differs between the thymus and extra-thymic AIRE expressing cells. To determine which proteins interact with AIRE in the periphery, lymph nodes from wild type mice will be analyzed using co-immunoprecipitation followed by tandem mass spectrometry. My hypothesis is that some of the important thymic coactivators, such as the transcriptional coactivator CBP [7], will interact with AIRE in eTACs, but other coactivators will differ. The ones that differ are likely responsible for the self-antigens that are uniquely expressed in eTACs. |
Future DirectionsMoving forward, it would be interesting to look at how self-reactive T-cell levels change when AIRE is knocked out in eTACs. Self-reactive T-cell levels could be measured in wild type vs eTAC AIRE knockout mice using flow cytometry. If there were more self-reactive T-cells in eTAC AIRE knockout mice than in the wild type, this would indicate that AIRE plays an important role in maintaining peripheral tolerance through its induction of self-antigen expression in eTACs.
|
Overall, learning more about AIRE's role in eTACs will help us better understand how AIRE helps maintain peripheral tolerance. Understanding the function of AIRE's domains, AIRE's role at different ages, and the proteins that interact with AIRE in eTACs will further our understanding of self-tolerance and autoimmune diseases. This knowledge can ultimately be applied to treating APECED and other autoimmune diseases.
References
[1] Kisand and Peterson. 2011. Autoimmune polyendocrinopathy candidiasis ectodermal dystrophy: known and novel aspects of the syndrome. Annals of the New York Academy of Science. http://onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.2011.06308.x/epdf
[2] National Organization for Rare Diseases (NORD). Hypoparathyroidism. https://rarediseases.org/rare-diseases/hypoparathyroidism/
[3] NIH Institute of Diabetes and Digestive and Kidney Disease. Adrenal Insufficiency and Addison's Disease. https://www.niddk.nih.gov/health-information/health-topics/endocrine/adrenal-insufficiency-addisons-disease/Pages/fact-sheet.aspx
[4] STRING. AIRE interaction network. http://string-db.org/cgi/network.pl?taskId=O8jn6ektBaKG
[5] Pitkanen et al. 2001. Subcellular Localization of the Autoimmune Regulator Protein. Journal of Biological Chemistry. Vol. 276, No. 22, pp. 19597–19602.
[6] The Jackson Laboratory. Life Span as a Biomarker. https://www.jax.org/research-and-faculty/research-labs/the-harrison-lab/gerontology/life-span-as-a-biomarker
[7] Fierabracci, Alessandra. 2011. Recent insights into the role and molecular mechanisms of the autoimmune regulator (AIRE) gene in autoimmunity. Autoimmunity Reviews. Vol. 10, p 137-143.
[2] National Organization for Rare Diseases (NORD). Hypoparathyroidism. https://rarediseases.org/rare-diseases/hypoparathyroidism/
[3] NIH Institute of Diabetes and Digestive and Kidney Disease. Adrenal Insufficiency and Addison's Disease. https://www.niddk.nih.gov/health-information/health-topics/endocrine/adrenal-insufficiency-addisons-disease/Pages/fact-sheet.aspx
[4] STRING. AIRE interaction network. http://string-db.org/cgi/network.pl?taskId=O8jn6ektBaKG
[5] Pitkanen et al. 2001. Subcellular Localization of the Autoimmune Regulator Protein. Journal of Biological Chemistry. Vol. 276, No. 22, pp. 19597–19602.
[6] The Jackson Laboratory. Life Span as a Biomarker. https://www.jax.org/research-and-faculty/research-labs/the-harrison-lab/gerontology/life-span-as-a-biomarker
[7] Fierabracci, Alessandra. 2011. Recent insights into the role and molecular mechanisms of the autoimmune regulator (AIRE) gene in autoimmunity. Autoimmunity Reviews. Vol. 10, p 137-143.
Final Presentation
|
| ||||||||||||
Presentation Drafts
| boles_presentationdraft1_2_19_17.pdf | |
| File Size: | 5776 kb |
| File Type: | |
| boles_presentationdraft1_2_21_17.pptx.zip | |
| File Size: | 5974 kb |
| File Type: | zip |
| boles_presentation_4_4_17.pptx.zip | |
| File Size: | 5220 kb |
| File Type: | zip |